P's exam 3 study guide
From Iusmhistology
[edit] Urinary 1

[edit] Cortex and Medulla
- Urine is produced by lobes which contain a single renal papillum which dumps urine into the pelvis which dumps into the ureter.


[edit] More on macrostructure
- Medullary pyramids are separated by renal columns of Bertin.

[edit] Uniferous tubule function

[edit] Uriniferous tubule layout and embryonic development
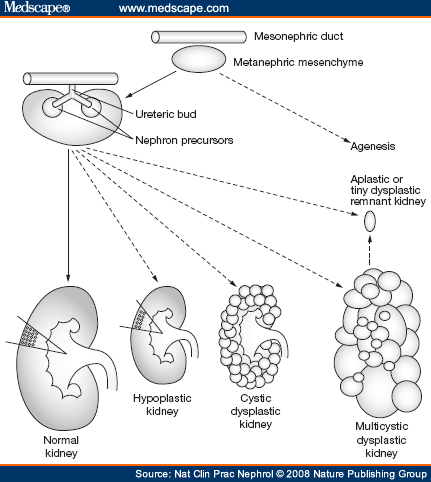
- The first form of a plasma filtering mechanism in the developing human embryo is called the mesonephric kidneys.
- Mesonephric kidneys reach their maximum size at 8 weeks and then undergo a large change and eventually are not kidney-like at all.
- Parts of the mesonephric kidneys persist in men to form:
- the efferent ductules,
- the epididymis,
- the ductus deferens, and
- the ejaculatory duct.
- so, everything after the straight tubules.
- The metanephros gives rise to the permanent kidneys.
- The metanephros contains the metanephric mesenchyme and the uritic bud.
- The uritic bud and the metanephric mesenchyme are both composed of epithelial cells.
- The uritic bud grows up into the nephrogenic mesoderm which is part of the metanephros.
[edit] Uritic bud and nephrogenic mesoderm interaction
- The uritic bud grows into the nephrogenic mesoderm to form the mature uriniferous tubules.
- The interaction between the uritic bud and the nephrogenic mesoderm is called reciprocal induction.
- As the uritic bud grows into the nephrogenic mesenchyme, the uritic bud is the primary epithelial cell tubule structure that will become the collecting duct.
- Recall that mesenchymal cells are connective tissue cells.
- Recall that mesenchyme looks like loose connective tissue with lots of spindly, undifferentiated cells within.
- Renal corpuscles develop along the length of the uritic bud (that is, the developing collecting duct) and therefore can originate from the tip of the uritic bud or from epithelium that develops along side the uritic bud.
- Renal corpuscle and nephron development from the tip of the uritic bud:
- At the tops of the uritic bud, mesenchymal cells of the nephrogenic mesenchyme condense and are induced to make a mesenchymal-epithelial transition (MET).
- Condensation includes proliferation
- These MET cells will become the epithelial cells of the glomerular capsule.
- The bud tip then expands to develop the PCT (proximal convoluted tuble), loop of Henle (LoH), and the DCT (distal convoluted tubule).
- The MET shifted cells of the early glomeruli recruit the formation of blood vessels that will become the glomerular capillaries.
- At the tops of the uritic bud, mesenchymal cells of the nephrogenic mesenchyme condense and are induced to make a mesenchymal-epithelial transition (MET).
- Renal corpuscle and nephron development adjacent to the uritic bud:
- Along side the uritic bud, epithelial tracts form as S-shaped or comma-shaped tubule structures.
- The tops of these se epithelial tracts will become the glomeruli and the length will become the PCT, LoH, and the DCT.
- The s-shaped buds from condensation, proliferation, and MET of mesenchymal cells will form the PCT, LoH, and DCT.


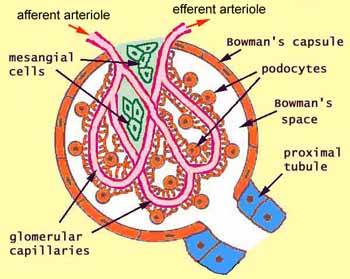
[edit] Renal corpuscle structure
- Note that podocytes are a type of epithelial cell.
- Capillaries are a type of endothelial cell.
- Within the capillaries as they develop within the glomerular tuft, there is connective tissue holding the capillaries in place.
- This connective tissue is called mesangium.
- The visceral bowmans capsule is made up of podocytes.
- Often there is pink material in the bowmans space; it is brush border from the proximal tubule that has washed backward during fixation.
- The distal convoluted tubule (which is, like the PCT, made up of epithelial cells) passes by the afferent arteriole along side the glomerulus.
- The DCT has specialized cells called 'macula densa cells on the surface that is closest to the afferent arteriole.
- Macula densa cells release signals PGE2 to cause the afferent arteriole to vasodilate and ATP to cause the afferent arteriole to constrict.
- Macula densa cells are more columnar, stain darker, and have rounder nuclei than the endothelail cells of the DCT.
- Juxtaglomerular cells (also called granular cells) are endothelial cells of the afferent arteriole that contain granules of renin.
- Granular cells (AKA juxtaglomerular cells) have a large, flattened nucleus, that is more prominent than the nucleus of lacis (extraglomerular mesangial) cells.
- Granular cells release their renin upon PGE2 binding their EP4 receptor.
- Recall that renin will activate angiotensinogen leading to angiotensin 2 and systemic vasodilation.
- Lacis cells (also called extraglomerular mesangial cells) hold the DCT, the afferent arteriole, and the glomerulus together.
- Extraglomerular mesangial cells may also have some functioning in modifying the signals released by the macula densa cells as they travel to the granular / endothelial cells of the afferent arteriole.
- Lacis cells (extraglomerular mesangial cells) are found between the macula densa cells and the afferent arteriole endothelial cells.
- Lacis cells have a lighter stain and less prominent nucleus as compared to granular (juxtaglomerular) cells.
- Extraglomerular mesangial cells are found between the convoluted capillaries, too, and serve to hold the loops in their structure.
- In this case, the mesangial cells are located within the basement membrane.
- Lacis cells can send processes into the lumen of the capillaries between the endothelial cells.





[edit] Forming a filter at the capillary-Bowman-space junction
- The filtrate must first get through the endothelium of the capillary, then through the basement membrane, and then through the feet of podocytes.
- The endothelium of glomerular capillaries is fenestrated without diaphragms to allow only very small proteins and smaller molecules through.
- The basal lamina restricts even the smallest proteins.
- There are three layers to the basal lamina (basement membrane) of the glomerulus.
- The three layers are probably only separate in slides as a result of processing, but they are still effective markers for pathology.
- The lamina rara extrna is farthest from the lumen of the capillary.
- The lamina rara interna is closest to the lumen of the capillary.
- The lamina densa is between the lamina externa and the lamina interna.
- These layers appear as a light-dark-light pattern in EM.
- Podocytes are a type of epithelial cell that provide the finest level of filtration (slit pore diaphragms) of the plasma as it crosses into the Bowman space.
- Water and small molecules pass freely into the Bowman space.
- It is still disputed what factors play the primary role in keeping proteins from entering the filtrate.
- Some say the anionic charge of the basement membrane, which would repel proteins which are generally negatively charged, is the primary factor that hinders protein passage.
- Others point to the podocyte processes and the important proteins that make up the processes (ZO1, nephrin, Neph1) as the primary protein-hindering mechanism.
- Nephrin seems to form a lattice between podocyte processes that would prevent proteins from passing into the bowman space.

- Recall that ZO1 is associated with tight junctions.
[edit] Mesangial cells
- Recall that mesangial cells reside between capillaries within the basement membrane.
- Recall that basement membranes are always made of type 4 collagen!
- Mesangial cells may modulate capillary blood flow.
- Mesangial cells may also act as phagocytes within the basement membrane of the glomerulus.
- Mesangial cells reaches out and cups each capillary around it.
- Mesangial matrix is made up of collagen, glycans, proteoglycans, etc.



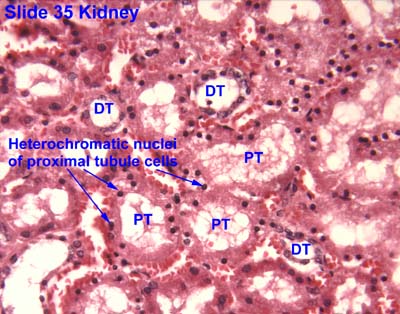
[edit] The proximal tubule
- The proximal tubule is characterized by being large, being eosinophilic (cuboidal, continuous, uniform), and having central nuclei.
- The proximal tubule demonstrates cells with brush border and basolateral membrane folding in order to increase its surface area.
- Note that during fixation, the brush border often sloughs off into the lumen.
- The proximal straight tubule continues through the outer stripe of the outer medulla.
- "Straight segments ... terminate at a remarkably uniform level ... that establishes the boundary between the inner and outer stripes of the outer ... medulla." per wikipedia
- Note that this is true for both cortical- and juxtamedullar glomeruli-derived proximal straight tubules.
[edit] Cell distinction along the PCT, LoH, and DCT
- Recall that the cells of the PCT, LoH, and DCT are all epithelial cells specialized for reabsorption and / or secretion.
- There are four regions that can be distinguished by cell morphology and characteristic: PCT / thick descending limb, thin descending / thin ascending, thick ascending / DCT, and the collecting duct.
- Note that the thick descending tubule is the same as the proximal straight tubule; the same goes for the distal region: distal straight tubule = thick ascending tubule.
[edit] Cells of the PCT and PST
- Note that the PST = proximal straight tubule = thick descending / proximal loop.
- There are only epithelial cells in the PCT and thick descending loop.
- Epithelium of the PCT is a simple squamous epithelium.
- the cells of the PCT and thick descending tubule are the only cells with a brush border.
- Cells of the PCT and thick descending tubule also have nuclei that are spaced far apart.
- PCT / thick descending tubule epithelial cells stain very pink.
[edit] Cells of the thin descending and thin ascending tubules
- There are only epithelial cells in the thin descending and ascending tubules.
- Recall that the descending loop is passively, highly permeable to water and solutes.
- Recall that the ascending loop is impermeable to water and actively secretes Na and Cl.
- The epithelial cells of the thin regions are thin cells that stain lightly.
- The nucleus of epithelial cells of the thin tubules is smaller than other nuclei of tubular epithelial cells.
[edit] Cells of the DST and DCT tubules
- The epithelium of the DCT and thick ascending tubule is thicker than the PCT and thick descending tubule.
- cell types in the thick ascending and DCT tubules: epithelial cells, macula densa cells, and principal cells, intercalated cells.
- Epithelial cells of the thick ascending tubule and DCT need lots of protein to facilitate ion transport and so it makes sense that thick ascending epithelium and DCT epithelium have lots of mitochondria.
- Epithelial cells of the thick ascending tubule and DCT have apical nuclei that bulge outward (perhaps because of the mt that are pushing them apically).
- Macula densa cells appear at the last part of the thick ascending tubule.
- The DCT is the first site of intercalated cells.
[edit] Differentiating PST and DST
- Thick descending epithelium stain darker than thick ascending epithelium.
- Thick descending epithelium has more basally located nuclei while ascending epithelium have apically located nulcei.
- Thick descending has a thicker wall than the thick ascending.
[edit] PCT versus PST and DCT versus DST identification
- Note that PST and PCT can be differentiated because they are never found in the same location: PCT is in the convoluted area and PST is only in the medullary ray area.
- Because the DCT and DST are both bound in the cortex, it is likely impossible to tell them apart (unless the structure in question runs right up next to a glomeruli and has macula densa at which point we know it is a DST).

[edit] Differentiationg PCT and DCT
- PCT and DCT can be distinguished by their stain and size:
- PCT epithelium has a brush border but DCT epithelium does not, though often the brush border is not preserved.
- PCT stains darker than DCT, though sometimes it can be the opposite, so good luck with that.
- PCT is made of larger cells than DCT (so with PCT you travel farther around the tubule before finding the next nucleus).

[edit] Cells of the collecting duct
- Epithelial cells of the collecting duct bulge into the lumen.
- Epithelial cells of the collecting duct have clear distinctions between each cell and have nuclei that do not bulge (like PCT / thick ascending tubule epithelial cells).
- Nuclei are more basal and irregularly shaped.
- Principal cells are hormonally controlled for water reabsorption and are the major site of potassium regulation.
- Principal cells absorb Na and secrete K.
- Principal cells are generally impermeable to water but can become water absorptive when ADH is present (think AQ2).
- Intercalated cells stain darkly, bulge a little into the lumen, have no brush border, have a more apical nucleus than principal cells, and are the site of pH regulation.
- There are three sections to the collecting duct: the connecting tubule and cortical collecting tubule, the outer medullary collecting tubule, and the inner collecting tubule.
- The two proximal sections (connecting duct / cortical collecting duct and the outer medullary collecting duct) have principal and interstitial cells; the inner medullary collecting duct has only principal cells.
- The inner medullary collecting duct is also called the papillary collecting duct.
- The last section of the inner medullary collecting duct is called the duct of Bellini.
[edit] Distinguishing regions of the kidney
- Note that thin segments of the LoH and DCT / PCT never occur in the same area so they can be used to determine the origin of a section.
- Thin loops of Henle are only found in the medulla.
- Recall that the thick proximal tubule terminates at the outer-inner stripe border of the medulla.
- Convoluted tubules are only found in the medulla.
- Thin loops of Henle are only found in the medulla.
- Distinguishing the medulla:
- The inner medulla has only asc / desc thin tubules and the collecting duct.
- The inner stripe of the outer medulla has asc / desc thin tubules, proximal / distal thick tubules, and the collecting duct.
- The outer stripe of the outer medulla has only thick tubules and collecting duct.
- There are no glomeruli in the medulla!
[edit] Urinary 2
[edit] Loop of Henle
- When the NaCl level is high, we want slow the filtrate flow rate so we have time to reabsorb all that valuable NaCl; therefore, when the NaCl level in the filtrate is high macula densa cells release ATP to constrict the afferent arteriole and decrease GFR.
- Conversely, very little NaCl in the filtrate at the macula densa means that the filtrate has had lots of time to have its NaCl reabsorbed so we can speed up GFR. In this case, macula densa cells release prostaglandins that cause renin release (and subsequently vasodilation) at the afferent arteriole.
[edit] More kidney superstructure

- Note that the ascending thick tubule is deeper than the descending thick tubule.
- Arcuate vessels follow the boundary of the cortex and medulla, giving off interlobular vessels that give off afferent arterioles and receive stellate vessels.
- The thick descending tubule = proximal straight tubule = pars recta.
- We can remove kidney stones through a surgery that pierces the cortex, enters a calyx, and uses a probe to grab / destroy the stone. Percutaneous nephroscopy.
[edit] Renal vasculature
- The order of renal blood flow: renal artery -> interlobar artery -> arcuate artery -> cortical radial artery (imagine these radiating outward from the arc; used to be called interlobular arteries) -> afferent arteriole -> glomerular capillaries -> efferent arteriole.
- The return route can start from two locations:
- Superficial and mid-cortical glomerulus: (from efferent arteriole) peritubular capillaries
- superficial peritubular capillaries return via stellate veins -> arcuate vein...
- deeper peritubular capillaries return via cortical radial vein -> arcuate vein...
- Juxtamedullary glomerulus: (from efferent arteriole) descending vasa recta -> ascending vasa recta -> arcuate vein...
- Superficial and mid-cortical glomerulus: (from efferent arteriole) peritubular capillaries
- Then both follow the same path away from their respective nephron: arcuate vein -> interlobar vein -> renal vein.

[edit] Cortex organization
- A renal lobule is a unit of renal tissue with medullary ray at the center with cortical radial vessels bounding it on the outsides.
- So medullary rays are the ascending and descending tubules that will run perpendicular to the capsule of the kidney.
- So, a cortical labyrinth is a collection of renal corpuscles with their associated medullary rays.
[edit] Juxtuloglomerular apparatus and the renin-angiotensin pathway
- Renin is released by granular cells (juxtaglomerrular cells).
- Angiotensin 2 causes systemic vasodilation.
- The apparatus contains the afferent and efferent arterioles, the macula densa, and the extraglomerular cells (lacis cells).
- There are also juxtaglomerular cells which are smooth muscle / endocrine cells.



[edit] Post-kidney urinary ultrastructure
- The calyces, pelvis, ureters, bladder, and uretra all have the same histological structure.
- The only exception is that the walls of the ureters become thicker as they continue.
- The calyces through bladder are transitional epithelium with a lamina propria and smooth muscle.
- Transitional epithelium allows these structures to change volume easily, which is most obviously important in the bladder.
- The lamina propria holds the cells together with connective tissue when changing volume.
- The smooth muscle allows contraction for movement of urine along the tract.
[edit] Transitional epithelium of the bladder
- Uroplakins can fold up like a pleat.
- A protein called uroplakin can be moved to the surface or removed from the surface via vesicular movement in order to increase or decrease surface area.
- Vesicles that contain uroplakin are called fusiform cytoplasmic vescicles.
- Uroplakin, as with all membrane proteins, is generated via the rER and golgi apparatus.
- Where uroplakin is on the surface, the membrane is thicker; there are thinner areas of membrane that are distict in EM of bladder epithelium.



[edit] Smooth muscle of the bladder
- Smooth muscle of the calyces, pelvis, and ureters are helical in pattern.
- Smooth muscle in the bladder is longitudinal and runs in all directions.
[edit] Endocrine histology
[edit] Describe the structural organization of the endocrine system
- Note that exocrine glands secrete onto an epithelial surface that is usually in the form of a duct whereas endocrine glands secrete into the blood stream.
- Furthermore, both exocrine glands and endocrine glands are usually on the outside of the basement membrane relative to the blood.
- Therefore, exocrine glands do not secrete across the basement membrane.
- However, endocrine glands often must secrete their hormones across the basement membrane.
[edit] Define components of the endocrine system
[edit] Describe the embryonic origin, histological organization, and hormone secretion of the endocrine system
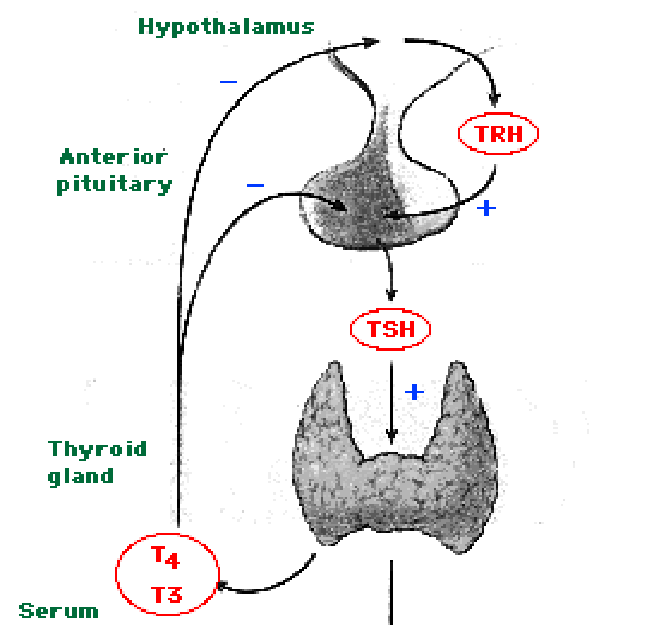
[edit] Origin, organization, and secretion of the hypothalamus
- The hypothalamus releases 5 hormones from three nuclei (dorsal medial, ventral medial, and infundibular nuclei):
- TRH which stimulates thyrotropes and mammotropes (lactotropes) of the anterior pituitary to release TSH and PRL.
- PIF (prolactin inhibitory factor, dopamine) which inhibits lactotropes (mammotropic cells) of the anterior pituitary from releasing PRL.
- SST (somatostatin) which inhibits somatotropes and thyrotopes of the anterior pituitary (adenohypophysis) to release GH and TSH.
- The hypothalamus also contains two more nuclei that produce two other hormones that are delivered directly to the posterior pituitary (neurohypophysis) through the axons of the neuron cells that produce the hormones.
- The supraoptic neucleus produces vasopressin (ADH, AVP) which acts on the collecting ducts of the kidney (think AQ2).
- The paraventricular nucleus produces oxytocin which acts on the mammary glands (myoepithelial cells) and uterus (smooth muscle cells, contractions).
[edit] Origin, organization, and secretion of the pituitary gland (hypophysis)
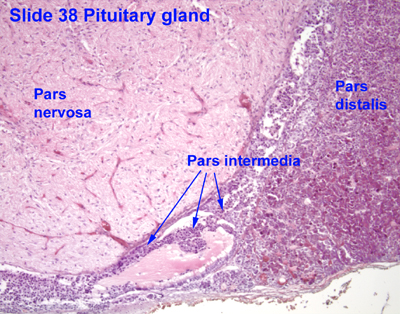
- The anterior pituitary consists of the pars distalis, the pars intermedia, and the pars tuberalis.
- The posterior pituitary (neurohypophysis) is made up of the pars nervosa and the median eminence.

- The neuroectoderm (floor of the diencephalon) grows caudally, forms a stalk, and remains attached to the brain tissue of origin.

[edit] Adenohypophysis (anterior pituitary)
- The pars distalis (anterior lobe):
- The pars distalis is composed of fibroblast generated reticular fibers that support hormone-generating epithelial cells and a rich bed of fenestrated capillaries.
- Cells of the pars distalis can be classified by the way the stain: basophilic, acidophilic, and chromophobes.
- Acidophilic cells: somatotropes and mammotropes (lactotropes).
- Basophilic cells: gonadotropes, croticotropes, and thyrotropes
- Chromophobic cells: stem cells, degranulated cells that would otherwise be chromophilic (see acidophilic and basophilic).
- Differentiating cell types is not possible with light microscope, only by trasmission electron microscopy can these hormone producing cells be differentiated.

- The pars tuberalis:
- Most cells of the pars tuberalis are basophilic.
- The pars intermeida:
- Colloid-filled cysts fill the pars intermedia.
[edit] Neurohypophysis (posterior pituitary)
- The neurohypophysis contains nerve cells and glial cells (pituicytes).
- The pars nervosa:
- The pars nervosa contains fibroblasts, pituicytes, mast cells and neurons.
- The neurons arise from the paraventricular and supraoptic neuclei where oxytocin and vasopressin are made, respectively.
- These neurons are atypical in that they do not synapse at their distal axons.
- The hormones released by these neurons are stored in granules (called Herring bodies or neurosecretory bodies) at the distal aspect of the axon.
- Herring bodies can be identified under light microscopy.
- The infundibular stalk
- The infundibular stalk, like the pars nervosa, contains atypical nerve axon endings that release hormones.
- The neurons of the infundibular stalk release their hormones into the hypothalamus-pituitary portal system and affect the cells of the anterior pituitary.
[edit] Pituitary portal system
- There are really 4 main components to the portal system: primary and secondary capillary beds, long veins and short veins.
- The primary capillary bed arises from the superior hypophyseal artery and resides around the median eminance.
- The long veins connect the primary capillary bed to the secondary capillary bed.
- The secondary capillary bed resides around the adenohypophysis.
- The inferior hypophyseal artery forms a capillary mesh at the neurohypophysis.
- The short veins connect the capillaries of the neurohypophysis to the secondary capillary bed of the adenohypophysis.
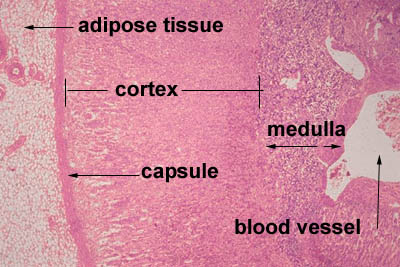
[edit] Origin, organization, and secretion of the Adrenal glands
- The outer shell is made of dense connective tissue that sends septa into the center of the organ as trabechulae.
- Cortex from mesoderm, medulla from neuro ectoderm.
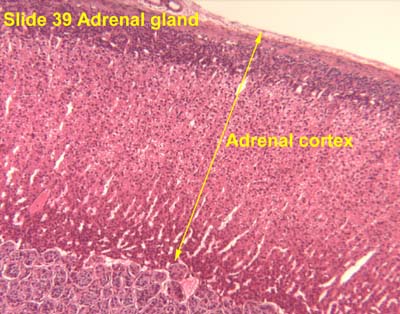
[edit] Adrenal cortex
- GomiFacoRea: glomerulus-mineralocorticoids, fasciculata-corticoids, reticularis-androgens.
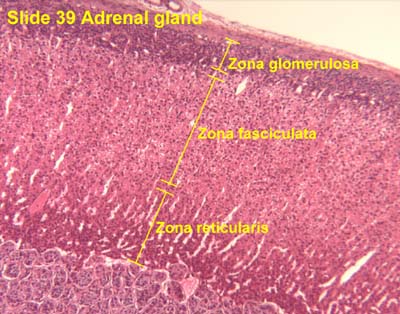
- The glomerulosa:
- The glomerulosa layer is characterized by closely-packed, arched chords of columnar or pyramidal cells surrounded by capillaries.
- The glomerulus can be differentiated from the capsule because of increased cellularity, prominent, circular nuclei, prominent arches, less connective tissue (which usually stains bright pink).
- The fasciculata:
- The fasciculata is characterized by long chords of polyhedral cellls and fenestrated capillaries.
- The fasciculata can be differentiated from the glomerulosa by distinct change in from short, bulbous cellular collections to long chord-like cellular collections.
- The reticularis:
- The reticularis can be differentiated from the fasciculata by less organized cellular collections, more eosinophilic staining (pinker, think about the granules of norepi and epi),



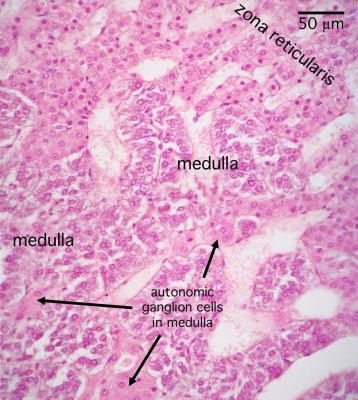
[edit] Adrenal medulla
- The medulla of the adrenal is composed of chromaffin cells which can be considered like post-ganglionic neurons.
- Chromaffin cells can either be norepinephrine producing or epinephrine producing and will have granules full of their labors.
- Norepinephrine-producing Chromaffin cells are found near medullary arteries.
- Epinephrine-producing Chromaffin are found near cortical sinuses.
- Cell density within the medulla is less than that of the cortex.



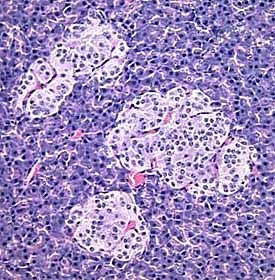
[edit] Origin, organization, and secretion of the Pancreas
- The endocrine portion of the pancreas arises from endodermal tissue near the bile duct.
- The notes also say that the endocrine protion arises from epithelium of the gut.
- There are four cell types in the endocrine islets of langerhans: beta, alpha, delta, and F / pp cells (by abundance).
- Delta cells make somatostatin.



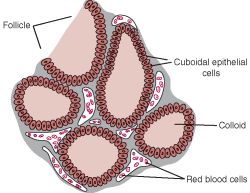
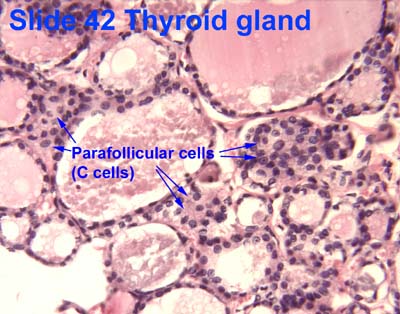
[edit] Origin, organization, and secretion of the Thyroid
- Within or between the follicles can be found C cells (parafollicular cells) which produce calcitonin.
- Production, storage, and release of thyroid hormones involves both endocrine and exocrine functions.
- Thyroglobulin made in rER, glycocylated in rER / golgi, and moved into the lumen.

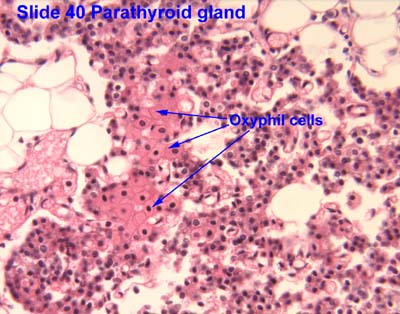
[edit] Origin, organization, and secretion of the Parathyroid glands
- The Parathyroid gland arises from the pharyngeal pouches.
- Like the adrenal glands, the parathyroid glands have a capsule with septa that run inward.
- The parathyroid gland is composed of two cell populations: chief cells and oxyphil cells.
- Chief cells:
- Chief cells contain eosinophilic granules of PTH.
- Note that regulation of chief cell PTH release is an inhibition of inhibitoin mechanism: when serum Ca levels decrease, fewer Ca-receptors bind Ca (the ligand) causing a decrease in intracellular signaling and an increase of PTH release.
- Oxyphil cells:
- The function of oxyphil cells is unknown; however, it is known that they arise during puberty.
- Oxyphil cells are larger than chief cells with an acidophilic cytoplasm and abnormally shaped mt.
- Oxyphil cells are often found in clusters at the center of the parathyroid gland or near the perimeter.



[edit] Primary hyperparathyroidism
- Primary hyperparathyroidism is a defect with the parathyroid itself causing an an elevation of PTH.
- Giving PTH intermittently to post-menopausal women is associated with decreased risk of bone fracture.
- continuous administration of PTH causes bone loss yet intermittent PTH administration causes increases in bone mass.
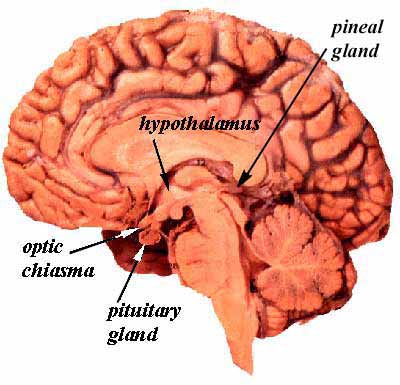
[edit] Origin, organization, and secretion of the Pineal gland
- The pineal gland arises from neuroectoderm from the floor of the diencephalon (just like the neurohypophysis).
- The pineal gland is pine-cone shaped and covered with connective tissue.
- This pine-cone shaped pineal gland is located in the posterior aspect of the third ventricle.
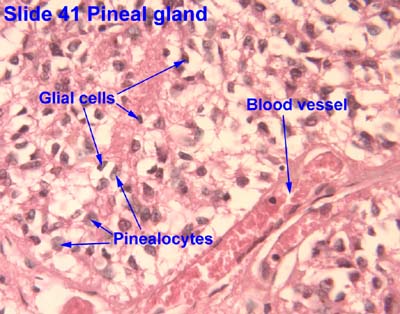
- The pineal gland contains pinealocytes, interstitial glial cells (like astrocytes).
- The pinealocytes produce melatonin and thus take part in daily rhythmicity.
- Rene Descarts explained human behavior and thought via the pineal gland because of its involvement in sensation, imagination, memory, and bodily movement.

[edit] Origin, organization, and secretion of the Diffuse Neuro-endocrine system
- Organs that have diffuse endocrine tissue include the heart, kidney, thymus, gut, and gonads.
[edit] Bone as an endcrine organ
- Two major signals are released by bone to affect physiology: FGF23 and uOCN.
- FGF23 is released by the bone and causes:
- Kidneys decrease phosphate (Pi) reabsorption resulting in decreased serium Pi.
- Kidneys decrease 1,25VitD activation resulting in decreased serum 1,25OH VitD and decreased Ca reabsorption.
- So FGF23 is generally an anti-bone-building signal.
- uOCN is released by the bone and causes:
- Pancreatic beta cells to increase insulin release resulting in decreased serum glucose.
- Adipocytes to increase adiponectin resulting in changes to glucose and fatty acid catabolism.
- Muscle to increase sensitivity to and uptake of glucose resulting in decreased serum glucose.
- So uOCN is generally a pro-growth-use-up-the-glucose signal.
- Bone also releases osteocalcin which has been shown to be associated with poor fertility when deficient.
[edit] Female reproductive histology
[edit] Ovarian cycle
[edit] Origin and fate of ovarian follicles
- Ovarian follicles are composed of a germ cell (oocyte) surrounded by supporting cells (follicular epithelial cells).
- Primordial germ cells originate from the yolk-sac (endoderm) and migrate to the genital ridge where the ovaries are developing.
[edit] Ovarian anatomy
- The cortex epithelium of the ovary is simple cuboidal epithelium.

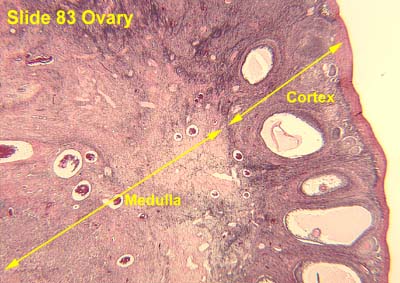
[edit] Ovarian follicle
- During the ovarian follicular phase mesenchymal cells will differentiate into theca cells to surround the follicle with an extra two layers.
- The endocrine layer of theca cells is called the theca interna; the vascular layer of theca cells is called the theca externa.
- The ovarian follicle has a specific anatomy of layers:
- Within the follicular cell population one may find a Call-Exner body which are collections of granulosa cell membrane with granulosa secretions within.
- The follicular cells are surrounded by a basement membrane (basal lamina).
- The basal lamina is surrounded by theca cells (from mesenchyme) which form two layers: theca interna and theca externa.




[edit] Stages of the ovarian follicle
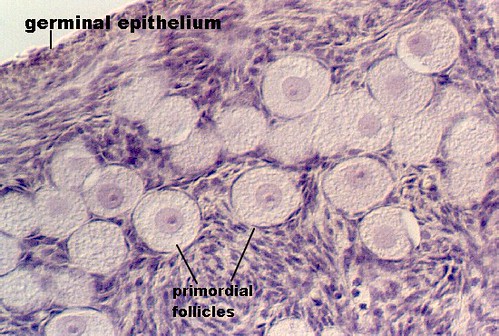
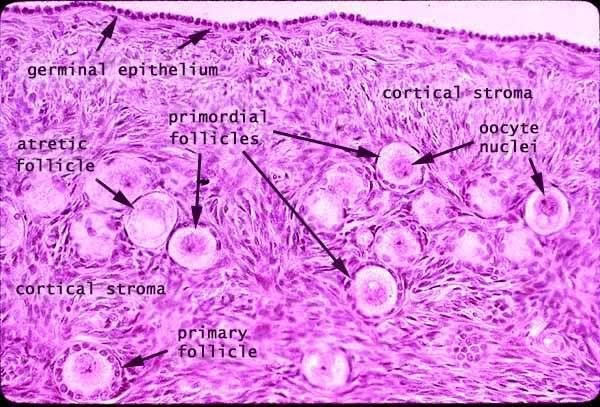
- Developing follicle:
- In the developing follicle, the follicular cells are cuboidal and have proliferated and differentiated into granulosa cells.
- Secondary follicle:
- The secondary (vesicular, antral) follicle is uniquely defined by a developing antrum and a theca externa.
- Granulosa cells secrete stroma-weakening factors to allow expansion of the follicle.
- A primary stroma-weakening factor is plasminogen-activator which converts plasminogen to plasmin (fibrinolysin, a trypsin-like enzyme) which cuts up fibrin.
- Granulosa cells secrete a meiosis-regulationg factors to inhibit movement from prophase 1 to metaphase 2 in the oocyte.
- It is in the secondary follicle stage (antral stage, vesicular stage) that the oocyte reaches its mature size.

- Mature follicle:
- The corona radiata is sometimes called the rim.
- The cumulus oophorus is sometimes called the stalk.
- Mature follicles are very large: can be over 1 cm!
- As a mature follicle, the oocyte progresses from prophase of meiosis 1 to metaphase of meiosis 2 and thus generates the first polar body.
- Note that having entered meiosis 2, the oocyte is called a secondary oocyte.
http://t0.gstatic.com/images?q=tbn:ANd9GcR_5ajvKIDofXIvPSq_8CEqUgDbjlsBRtYNqKEDaGYemdfQoqPAtQ

- Deciding on which type of follicle you're observing:
- flattened follicular cells: primordial
- cuboidal folliclar cells (with a layer on either side--zona pellucida or basal lamina): developing
- antrum / fluid: secondary follicle
- no way to distinguish secondary from mature.

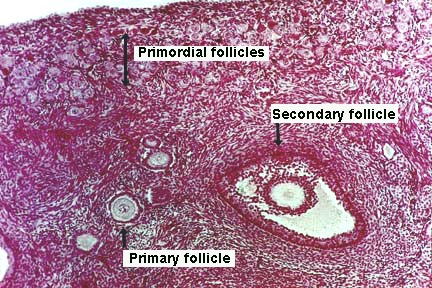
- Here is a good image (though it does not show a mature follicle):



[edit] Endocrine regulation of follicle maturation
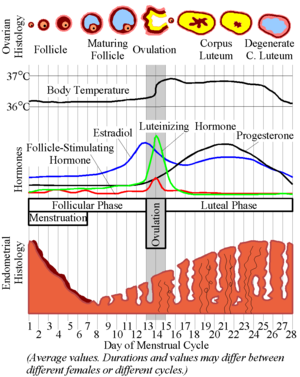
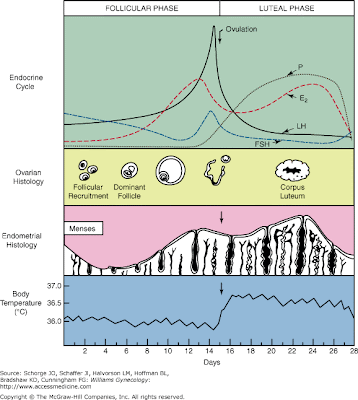
- Increased estrogen inhibits FSH release at the pituitary thus stopping the growth of the follicle and allowing ovulation.
- Increased estrogen stimulates LH release at the pituitary thus commencing ovulation.
- Recall these classic images:

[edit] Ovulation
- As the granulosa cells of the secondary and mature follicle produce more and more estrogen, more and more LH is released from the anterior pituitary gland (adenohypophysis, pars distalis).
- Ovulation events include:
- Breakdown of the cumulus oophorus, thus the oocyte floats freely in the antrum and follicular fluid.
- Weakening of the ovarian stroma:
- Proteolytic enzymes like collagenase disrupt the stromal connective tissue.
- Granulosa cell connections weaken
- Local ischemia causes a pale spot on the surface of the ovary called a stigma.
- Follicular wall ruptures releaseing an oocyte with the corona radiata and zona pellucida surrounding.
[edit] The corpus luteum
- LH causes granulosa cells to become granulosa lutein cells and theca cells to become theca leutein cells.
- Granulosa lutein cells develop the morphology of a secretory cell and actively produce progesterone.
- Note that the production of progesterone by the granulosa lutein cells is necessary for implantation of the embryo.
- LH causes theca cells to become theca lutein cells.
- If pregnancy does not occur, the corpus luteum is called the corpus luteum of menstruation.
- Therefore, progesterone from the corpus luteum is self limiting.
- That is, the corpus luteum will bring about self-demise via progesterioen inhibition of pituitary-LH unless chorionic gonadotropin is generated by the placenta.
- If pregnancy occurs, the corpus luteum is called the corpus luteum of pregnancy.
- Recall that trophoblasts of the placenta produce chorionic gonadotropin which maintains the corpus luteum (even though LH drops because of high progesterone levels).
- hCG is the hormone used to test for pregnancy.
- Granulosa cells of the corpus luteum of pregnancy produce relaxin which has a smooth-muscle relaxing effect (histo says "during parturition", wikipedia says "during gestation").
- Relaxin opposes the pro-parturition actions of oxytocin; that is, it keeps the smooth muscle of the uterus relaxed.
- Relaxin targets the fibrocartilage of the pubic symphysis to increase articulation.
- Note that physio notes say that the role of relaxin in pregnancy is unclear.

[edit] Follicular atresia
- Follicular atresia generates a long-lasting, scar-tissue structure called the corpus albicans.
- The zona pellucida remains (bright pink) and a wavy line (the basement membrane, called the glassy membrane).

[edit] The uterine tubes
- The uterine tubes are muscular tubes that extend from the ovary on the posterolateral wall of the abdomen to the medioventral aspect of the abdomen and the lateral aspect of the uterus.
- As the oviducts progresses distally, there are fewer involdings.


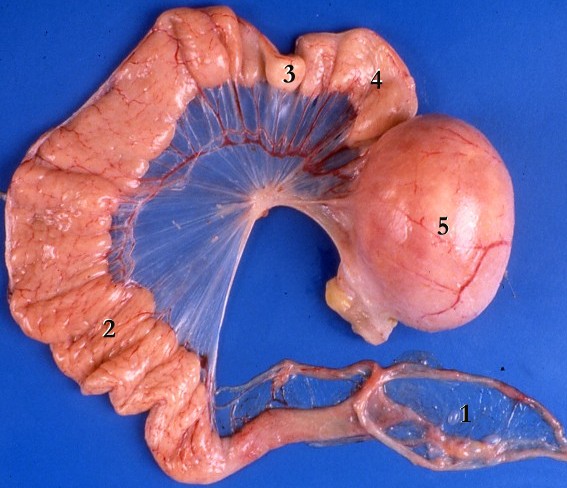
http://www.jci.org/articles/view/29424/files/JCI0629424.f1/medium
[edit] Layers of the oviduct
- Like other epithelial tracts there are four major layers to the oviduct (from inner to outer): mucosa, lamina propria, muscularis, and serosa.
- Secretions from the oviduct promote sperm activation.
Does this refer to capacitation?
- Mucosa of the oviduct:
- The mucosa of the oviduct is comprised of columnar, ciliated epithelial cells.
- These columnar cells are secretory and are called Peg cells.
- Estrogen (from the corpus luteum) increases the height of the columnar cells.
- Progesterone (from the corpus luteum) increases the ciliary action of the columnar epithelial cells of the mucosa.




- Lamina propria:
- The lamina propria of the oviduct is highly vascularized.

- Muscularis:
- As with so many muscularis layers, there is an inner circular and outer longitudinal layer.
- The two layers of the muscularis are interwoven.

- Serosa:
- The serosa is a true serosa because it is lined with mesothelium.



[edit] The uterus
[edit] Layers of the uterine wall
- Like the oviducts and other epithelial tracts, there are four tissue-type layers to the uterus which make up three functional layers of the uterus.
- The epimetrium is composed of serosa and adventitia and is a form of mesothelium as one would expect to cover surface of organs that faces the inside of the abdomenal cavity.
- Note that the cervix is histologically distinct from the rest of the uterus; we will revisit this.
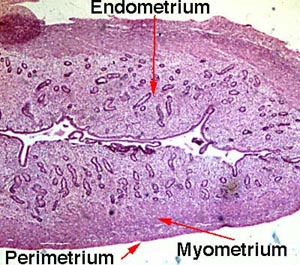



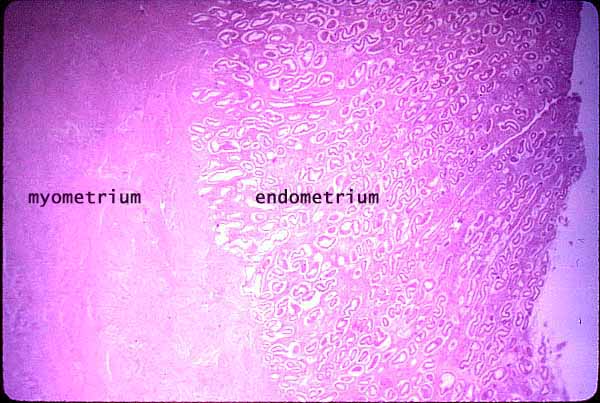

[edit] The uterine cycle
- From physio notes:
- The proliferative stage is characterized by endometrium hypertrophy and formation of spiral arteries.
- So as the ovary is maturing its follicle, the uterus is regenerating it's surface (where the egg will implant) and increasing vascular access to the surface.
- The secretory stage is characterized by coiling of glands, secretion of mucus, tortuous arteries, and peak thickness of the endometrium.
- So, as the ovary has shed an ovum and is now increasing hormone production via the corpus luteum, the uterus is using glands and arteries of the uterus to modify the uterine microenvironment to the optimal conditions for egg implantation.
- The ischemic stage is characterized by arterial constriction, decreased blood flow, and increased prostaglandins.
- So as the ovary has reached its lowest levels of hormone production, the uterus is decreasing nutrition to the endometrium and allowing the mucosa to undergo necrosis by ischemia.
- The menstrual stage is characterized by desquamation of the endometrium.
- So as the ovary has reached its lowest levels of hormone production, the uterus is shedding its endometrium.
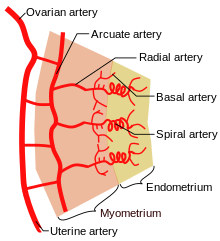
[edit] Uterine vasculature
- The uterus is supplied by arcuate arteries that run along the myometrium layer and by radial arteries that cross into the endometrium.
- The radial arteries give off straight (basal) arteries that supply the endometrium basalis.
- Note that the endometrium is divided into two layers: the endometrium basalis is a constant, mostly unchanging layer while the endometrium functionalis cycles through generation (proliferation) and shedding.
- There is no structural marker to distinguish between the basalis and the functionalis of the endometrium.
- Spiral (coiled) arteries are heavily muscular, generated during the endometrial cycle, and bridge the radial arteries into the endometrial functionalis.
- Of special note are the vascular structures nearest the lumen of the uterus called lacunae.
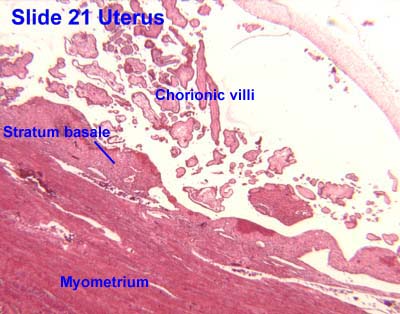
[edit] Uterine endometrium
- The endometrial mucosa contains uterine glands.
- Uterine glands are tubular with many branches.
- Uterine glands contain both ciliated and non-ciliated cells.

[edit] Histological changes in the uterine cycle
- The phases are divided over approximately 28 days: menstruation (days 1-5), proliferation (6-15), secretion (16-17), ischmia (18-28).
- Menstruation:
- Note that the base of the uterine glands remain visible in the endometrium basalis.

- Proliferative stage:
- The proliferative stage is driven by estrogen produced by the developing follicle.
- In addition to gland coiling, spiral arteries develop in the thickening endometrium.
- Cells of the proliferative endometrium accumulate glycogen.
- Secretory stage:
- The secretory stage is driven by progesterone from the corpus luteum.
- The secretory stage is characterized by release of glycoprotein-rich products, swelling and torture of the glands and spiral arteries, and accumulation of fluid in the stroma of the endometrium.
- Ischemia:
- The ischemic stage is characterized by constriction of the coiled arteries, stromal fluid loss, and lymphocyte / macrophage cell invasion.
- When the corpus luteum degenerates and progesterone levels drop, local prostaglandins are released into the endometrium, the vessels constrict, and blood flow is arrested causing ischemia.
- The coiled arteries dilate and constrict intermittently which causes ischemia, cell lysis, a weakened stroma, bursting vessles, and debridement of the functionalis.
- The arteries both restrict oxygen (constriction) to cause cell death but also to flush away the dead tissue (dilation).
- The ischemic stage is characterized by constriction of the coiled arteries, stromal fluid loss, and lymphocyte / macrophage cell invasion.
[edit] Uterine cervix
- As mentioned before, the cervix is histologically distinct from the rest of the uterus.
- The cervical myometrium:
- The cervical myometrium has less smooth muscle and abundant collagenous connective tissue with elastic fibers.
- The cervical endometrium:
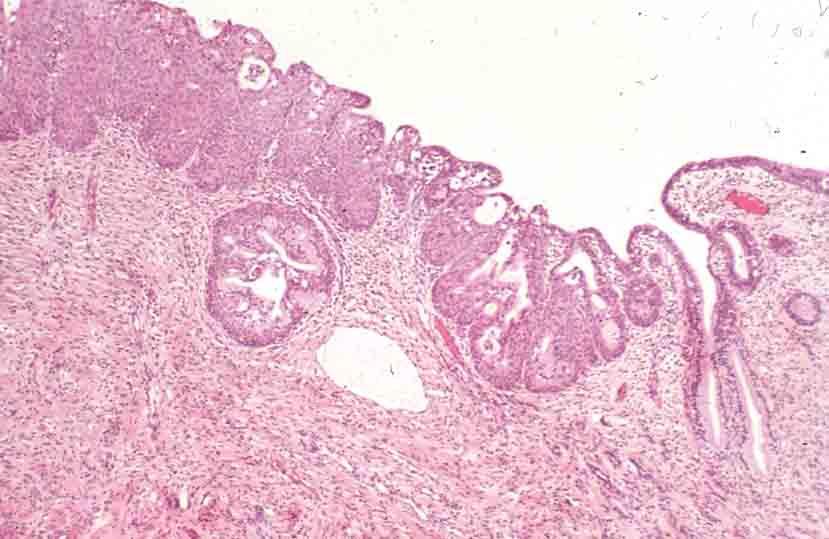
- The cervical endometrium has denser stroma, simple columnar epithelium, branched, dilated, cyst-forming glands, and longitudinal mucosal folds called plicae (plicae palmatae).
- Cervical glands can form cysts called Nabothian cysts.
- The cervical endometrium has denser stroma, simple columnar epithelium, branched, dilated, cyst-forming glands, and longitudinal mucosal folds called plicae (plicae palmatae).
- The cervical mucus:
- Mid way through the cycle (think ovulation and sperm-friend environment) the mucus is watery, contain lysozyme (bacterioalcidal), and promotes sperm motility.
- This sperm-friendly mucus is estrogen-stimulated.
- Late in the uterine cycle (think corpus luteum and potential implantation) the mucus is viscous and progesterone-stimulated.
- During pregnancy the mucus is particularly thick (think lots of progesterone) and thus protective of the fetus.
- One may look for the loss of this dense mucus plug as a sign that parturition is commencing.
- Mid way through the cycle (think ovulation and sperm-friend environment) the mucus is watery, contain lysozyme (bacterioalcidal), and promotes sperm motility.
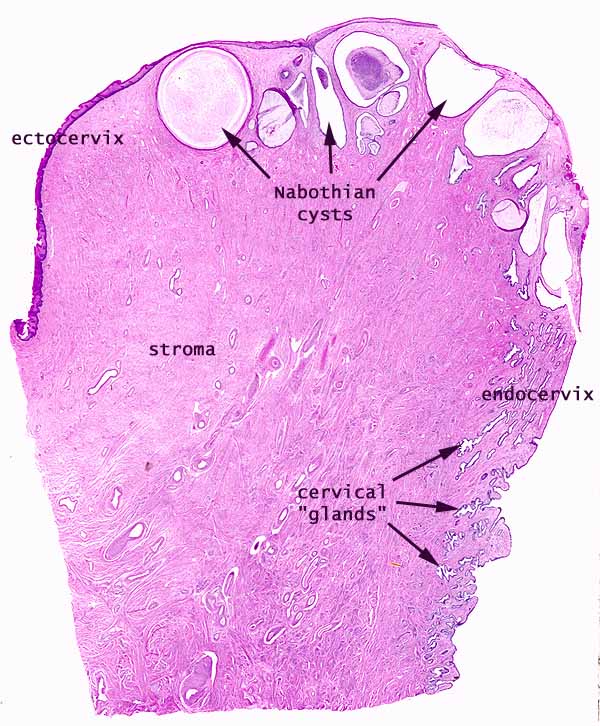
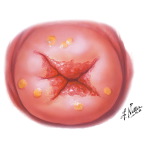
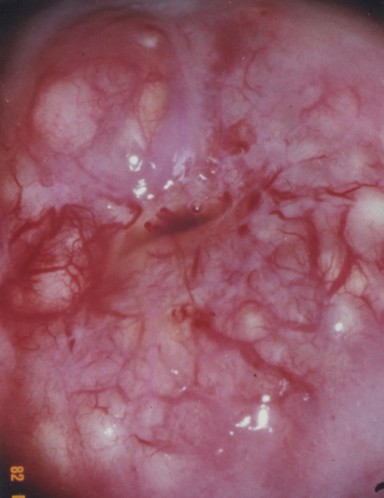


[edit] The ectocervix
- The ectocervix is also called the portio vaginalis.
- At the ectocervix the epithelium changes from columnar (cervix) to stratified squamous (vagina) abruptly.
- Normal ecotcervix:


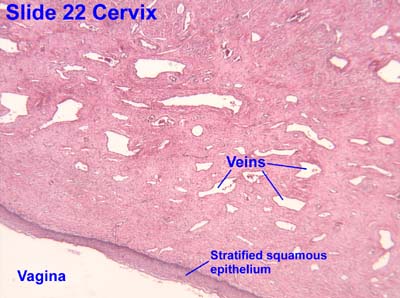
[edit] Vagina
- The mucosa is characterized by stratified, squamous, non-keratinized epithelium.
- The epithelial cells of the vagina--like those of the uterus--accumulate glycogen upon estrogen signaling.
- The vaginal lamina propria has no glands, patches of lymphocytes, and can have folds.
- Recall, however, that the uterus does have glands in the lamina propria.
- The vaginal muscularis has interlacing bundles of smooth muscle.

.jpg)
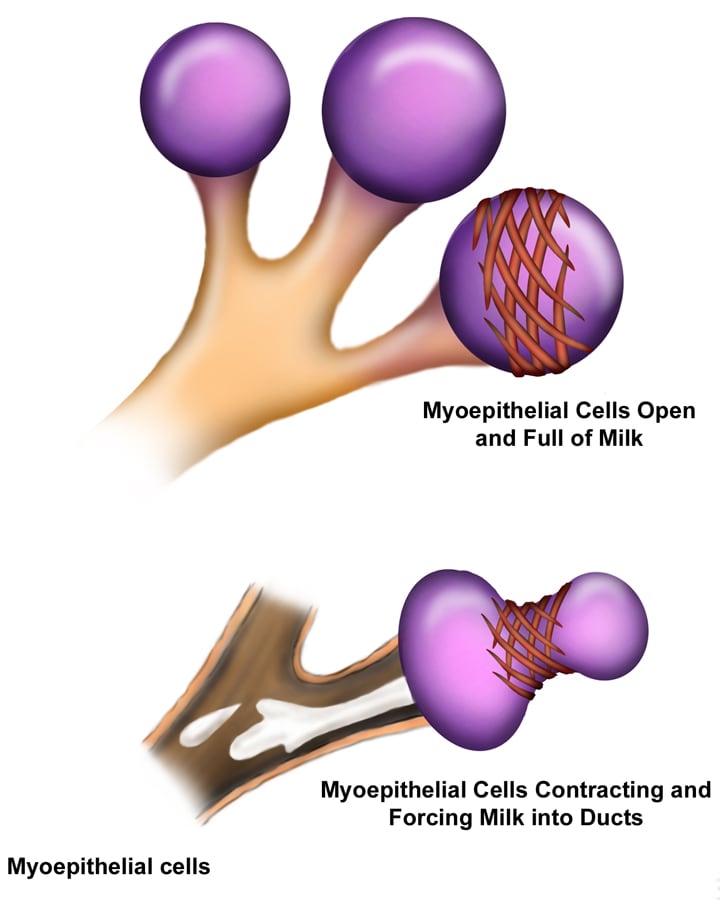
[edit] Mammary glands
- The mammary glands are made of a compound tubuloalveolar system; that is, there are alveoli that take part in the secretory component and there are ducts that take part in the transport component.
- A group of about 20 glands forms a mammary lobule.
- Secretory component:
- Milk is generated by cuboidal epithelial cells arranged in alveoli.
- The myoepithelial cells arise from the cuboidal epithelial cells.
- Plasma cells are found in and around the alveoli in order to generate IgA.
- Mammary ducts:
- The epithelium that lines the ductule system is stratified cuboidal.
- There is a significant amount of smooth muscle between the ducts and sinuses.
- Most breast cancers arise from lactiferous duct cells.






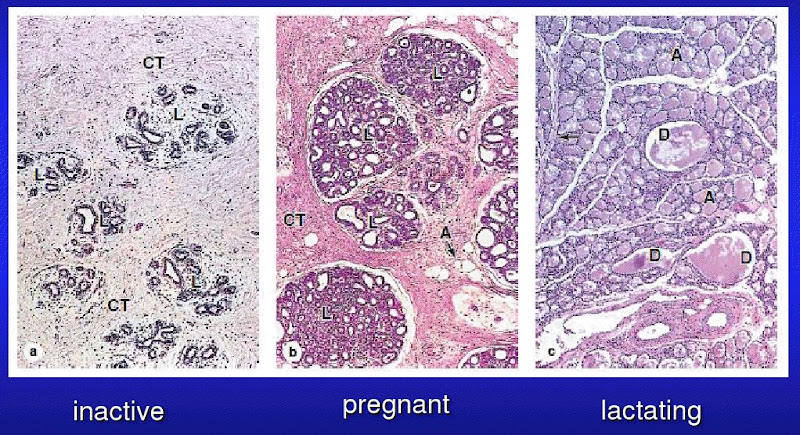
[edit] Mammary glands at puberty
- When estrogen increases at puberty (and prolactin is present), alveolar buds develop and regress with each ovarian cycle.
[edit] Mammary glands in pregnancy
- Upon pregnancy, estrogen is found at high levels along with prolactin, placental lactogen, and progesterone and therefore the alveolar ducts and alveoli fully develop.
- Lactogenesis is regulated by estrogen, progesterone, and prolactin.
- This makes sense because estrogen and progesterone increase throughout pregnancy and once progesterone and estrogen drop (at parturition), prolactin has its most potent effect.
- Galactogenesis is maintained by prolactin and oxytocin.

[edit] Breast milk
- The first milk generated is called colostrum; colostrum is lactoprotein- and immunoglobulin- rich and lipid-deficient.
- Lactation can generate 1100 to 2100 ml every day.
- Note that physio said 800-1200 ml / day.
- These cuboidal epithelial cells use several secretion mechanisms to release their products.
- Merocrine secretion is used to secrete casein, alpha-lactalbumin, and PTH-RP (protein, basically).
- Apocrine secretion is used to secret TAGs and cholesterol.
- Exocytosis is used to secrete lactose.
- Transcytosis (from adjacent plasma cells) is used to secrete dimeric IgA.
[edit] Regulation of milk let-down
- Note that afferent CNS signals also stimulate dopamine inhibition such that prolactin is increased which stimulates the cuboidal epithelial cells of the alveoli to increase milk production.
- Note that multiple neuroendocrine factors have been found besides oxytocin to relax the smooth muscle sphincter between the lactiferous ducts and the lactiferous sinuses.
[edit] Summary
- Estrogens increase as the follicle develops and:
- inhibit FSH at the pituitary
- stimulate LH at the pituitary
- LH promotes corpus luteum formation
- Corpus luteum produces progesterone and estrogen
- Estrogen causes uterine proliferation phase
- Progesterone causes uterine secretory phase
- Progesterone inhibits LH
[edit] Male reproductive
[edit] Anatomy review
- The mediastinum testis is where the vessels (blood and lymphatics), nerves, and efferent duct enter and exit the testis.
- Note that the mediastinum testis is connective tissue while rete testis is a collecting tubule tissue.
- The tunica propria is the outer wall of the seminiferous tubule and is made of smooth muscle and fibroblasts.
[edit] Seminiferous epithelium

[edit] Spermatogenesis
- Primary spermatocytes are in the prophase of meiosis 1 and stick around for 20 days.
- Secondary spermatocytes are relatively short-lived.
- Cells of the basal compartment: type A and type B spermatogonia, primary spermatocytes
- Cells of the adlumenal compartment: secondary spermatocytes, spermatids, spermatozoa
- Cells and processes: type A spermatogonia undergo mitosis to become ... type B spermatogonia undergo mitosis (and differentiation) to become ... primary spermatocytes undergo meiosis 1 ... secondary spermatocytes undergo meiosis 2 ... spermatids undergo morphologic modification (differentiation) ... spermatozoa.
- Spermatocytogenesis includes all the steps that generate an increasing number of cells (that is, type A spermatogonia through generation of secondary spermatocytes); this makes sense because of the name "cyto" = cell and genesis = "origin of".
- Spermiogenesis is the converse of spermatocytogenesis: spermeiogenesis is the maturation of existing cells into spermatozoa (from the secondary spermatocyte stage to the spermatozoa stage).



[edit] Mitosis and Meiosis
[edit] Spermiogenesis
- Spermiogenesis is characterized by morphological changes to the spermatid, that is the specialization / differentiation of the spermatid into the spermatozoa:
- Golgi phase:
- The enzymes like hyaluronidase and trypsin-like protease accumulate at one pole of the nucleus in a vesicle (which will become the acrosome).
- Acrosomal phase:
- The cell rotates such that the axoneme faces the lumen.
- Maturation phase:
- The maturation phase is characterized by motile apparatus development and the capacity to fertilize.
- Note that during the maturation phase, the spermatids are not yet motile or fertile.

[edit] Sertoli cells
- Sertoli cells are characterized by being tall, columnar epithelial cells with a large, indented euchromatic nucleus, and lots of eosinophilic cytoplasm.
- Neighboring Sertoli cells within a region have gap junctions which suggest that Sertoli cells are coordinated within their region.
[edit] Blood testis barrier
- Sertoli cells of the seminiferous epithelium form tight junctions between one another to keep immunoglobulins in the blood from entering the lumen of the tubule.
- Note that these tight junctions of the Sertoli cells are on the lumenal side of the spermatogonia.
- These tight junctions define the two compartments: basal compartment and adlumenal compartment.
[edit] Sertoli function stimulated by FSH
- Recall that the anterior pituitary releases FSH which binds to the FSH receptor on Sertoli cells.
- FSH signaling on Sertoli cells causes phagocytic activity and production of several secretions.
- Secretions of the Sertoli cells:
- Activin and Inhibin: stimulate and inhibit the anterior pituitary cells to release FSH.
- Androgen binding protein (ABP): secreted into the lumen of the seminiferous tubule, binds up and concentrates testosterone.
- Tubular fluid: lubrication.
[edit] Leydig cells
- Note that Leydig cells do not secrete activin or inhibin.
- Leydig cells are found in clusters in the peritubular interstitium of the testis, between the seminiferous tubules.
- Recall that Leydig cells are often found near capillaries.
- Leydig cells are characterized by eiosinophilicism, lots of sER, mt with tubular cristae, and a lack of secretory vesicles.
- Regarding lots of sER, recall that steroids are generated in sER.
- Regarding a lack of secretory vesicles recall that steroids can pass directly through the membrane and therefore need not vesicular secretion.
- By transmission EM, one can also discern crystalline inclusions.
- Leydig cells are "transiently" active during development and then initiate full activity at puberty.
[edit] Segments of the male reproductive tract
- Seminiferous tubules -> tubuli recti (straight tubules) -> rete testis -> efferent ductules -> epididymal ducts -> ductus deferens (vas deferens) -> ejaculatory duct -> prostatic uretral -> membraneous urethra -> penile urethra.
Where do basal cells and principal cells start?
- Straight tubule (tubuli recti)
- Contributes to fluid
- Cuboidal to columnar
- Contains Sertoli cells
- Rete testis
- Contributues to fluid
- Has an irregular epithelial morphology: squamous, cuboidal, columnar.
- Has a fibrous stroma
- Efferent ductule
- Like the rete testis, the efferent ductule has an irregular epithelial morphology and also has ciliated, pseudostratified cells.
- These ciliated pseudostratified cells are called principal cells and are found as clusters of columnar cells surrounded by short cells.
- These cells are the only ciliated cells of the male genital tract; it even makes a bit of sense that these are ciliated because they are passing through a sturdy connective tissue structure.
- Like the rete testis, the efferent ductule has an irregular epithelial morphology and also has ciliated, pseudostratified cells.
- Ductus epididymis
- The ductus epididymis is characterized by an pseudostratifed columnar epithelium, stereocilia, and a prominent muscularis layer.
- Basal cells are regenerative.
- Principal cells are secretory / absorptive.
- The ductus epididymis has a prominent muscularis layer that thickens distally and changes from just a circular layer to an inner circular and outer longitudinal layer.
- The functions of the ductus epididymis: absorb 90% of the tubular fluid, secrete factors that mature and capacitate sperm, and phagocytize cellular debris.
- Ductus deferens (vas deferens)
- The ductus deferens is characterized by a pseudostratified columnar epithelium, abundant sympathetic innervation, and three layers to the muscularis.
- The ductus deferens is pseudostratified (like the efferent ductule and ductus epididymis) and has stereocilia (like the ductus epididymis).
- The muscularis has now added an inner longitudinal muscle layer to the ductus epididymis's inner circular and outer longitudinal.
- The sympathetic innervation will be important for ejaculation which is facilitated by contraction of the ductus deferens's muscularis.
- Recall that Point and Shooting require Parasympathetic and Sympathetic innervation.
- Ejaculatory duct
- Membranous urethra
- The membranous urethra is characterized by a pseudostratified epithelium that transitions to a stratified columnar epithelium.
- Penile urethra
- The penile urethra is characterized by a pseudostratified epithelium that transitions to a stratified columnar epithelium, perhaps continuing on to stratified squamous epithelium at the top.
- The penile urethra is also characterized by the presence of urethral glands which are clusters of mucus cells in the mucosa.
| Section | Epithelium | Appendages | Muscularis |
|---|---|---|---|
| Seminiferous tubule | stratified | none | none |
| Tubuli recti | cuboidal -> columnar | none | none |
| Rete testis | irregular: squamous, cuboidal, columnar | none | none |
| Efferent ductule | pseudostratified | cilia | none |
| Ductus epididymis | pseudostratified | sterocilia | circular -> circular (inner) + longitudinal |
| Ductus deferens | pseudostratified columnar | stereocilia | longit (inner) + circular + longit |
| Ejaculatory duct | pseudostratified columnar | ? | ? |
| Prostatic urethra | pseudostratified -> simple columnar | ||
| Membranous urethra | pseudostratified -> stratified columnar | none | longit (inner) + circular + longit |
| Penile urethra | pseudostratified -> stratified columnar -> stratified squamous | none | longit (inner) + circular + longit |
[edit] Accessory glands of the male reproductive tract
[edit] Seminal vesicles
- Seminal secretions are rich in fructose (for energy), prostaglandins, amino acids, and ascorbic acid.
- The seminal vesicle has plenty of smooth muscle with which to force this secretion out into the ejaculatory duct.
- Seminal vesicle function and growth is mediated by testosterone.
- The vesicles are blind-ended pouches near the prostate and ductus deferens.
- Seminal vesicles are lined with pseudostratified columnar epithelium.
- This makes sense because they pump semen out into the GU tract at the ductus deferens and ejaculatory duct, both of which are pseudostratified.
- The seminal vesicles have mucosal arches which are highly folded, convoluted walls.
[edit] Prostate
- The prostate generates a secretion that becomes part of the semen and serves to condition the environment of the famale GU tract.
- The fribromuscular stroma surrounding the prostate is important for proper discharge of prostatic secretions during ejaculation.
- Zinc inhibits macrophage activity.
- Fibrinolysin inhibits clot formation in the uterus.
- The prostate as a gland has alveoli connected via tubules; the tubules converge to generate a group of ducts that dump into the urethral crest.
- The urethral crest receives the ejaculatory ducts and becomes the prostatic urethra.
- The epithelium within the gland is irregular: pseudostratified to simple columnar.
- Corpora amylacea is a concentration of glycoprotein-rich secretion in the lumen of the gland that stains eosinophilic (because of the "protein-rich" part).
- The prostate has three concentric-like zones: central zone, transitional zone, and peripheral zone.
- The central zone of the prostate is the most medial and contains primarily periurethral mucosal glands.
- The central zone often stains the lightest of the zones.
- The transitional zone of the prostate is the middle zone and contains periurethral submucosal glands.
- Note that the central and transitional zones are most commonly associated with benign prostatic hypertrophy (BPH).
- The peripheral zone of the prostate is the outer zone, contains the main glands, is called the prostate proper, and is commonly involved with malignancies.
- The peripheral zone is often involved with prostate cancer.
[edit] Bulbourethral glands
- The bulbourethral glands produce a secretion for lubricating the male GU tract for ejaculation.
- The secretion of the bulburethral gland is clear and viscous.
[edit] Ejaculation sequence
- Ejaculation has a particular series of events regarding all these secretions:
- Bulbourethral glands discharge to lubricate.
- Prostate releases contents (via contraction of the fibromuscular stroma).
- Recall that the prostate's secretions serve to condition the female GU tract.
- The ductus deferens receives sympathetic stimulation to contract its muscularis (1->2 layers), thus pushing spermatozoa into the urethra.
- Seminal vesicles discharge their contents thus clearing the urethra by pushing semen distally.
- ID straight tubules by the presence of Sertoli cells.
- Ductus epididymis has very tall cells with cilia.
- Ductus deferens ID by stereocilia and three layers of muscle.
- Seminal gland: mucosal arches
- Prostate is ID'd by corpora amylacea (a concentration of glycoprotein-rich secretion in the lumen of the gland that stains eosinophilic).
[edit] Skin
[edit] Describe the basic histological structure of the skin


[edit] Identify the cell layers that constitute the epidermis
- Also, the granulosa and corenum layers are thicker in "thick skin".


[edit] Stratum basale
- The cells of the basal layer are connected to the basement membrane via hemidesmosomes.
/Integument/Thick%20Skin%20-%20Tutorial/F%20-%20Thick%20Skin%2040X-3-Epidermal%20Layers.jpg)
[edit] Stratum spinosa



[edit] Stratum granulosmum
- The granule-containing cells of the granulosum contain filaggrin, and intermediat filaments that help to form the tonofibrils along with keratin.
- Note that these granules are called keratohyline granules.
- Kertohyline granules have no membrane.
- The cells of the granulosum are flattened polygonal cells--found in 3 to 5 layers.
- Just like "lamellae" in chloroblasts, lamellar granules have a sort of stacked-bags look
- Labellar graunules cannot be seen in light microscopy.


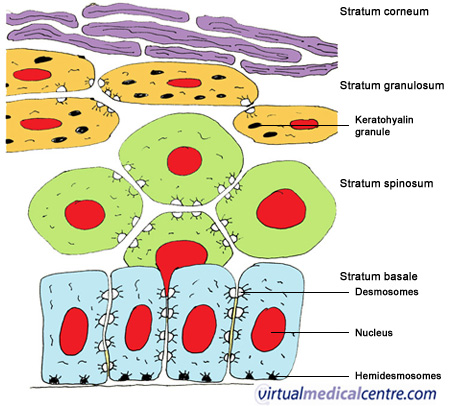
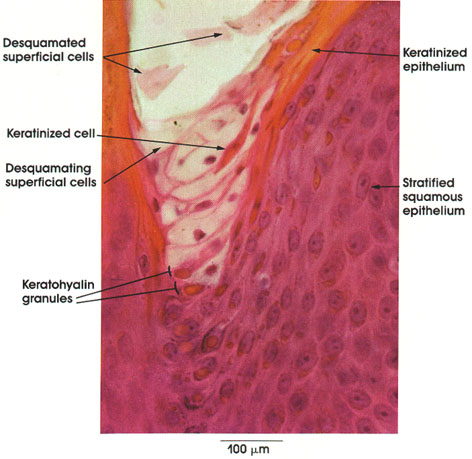
[edit] Stratum lucidum
- The lucidum (ironically) stains darkly.
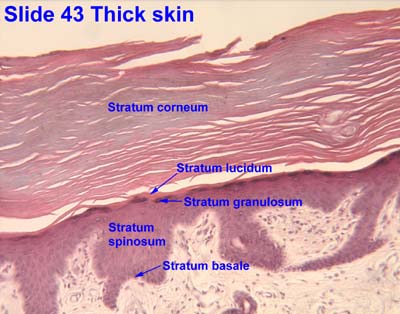
[edit] Stratum corneum
[edit] Dermo-epidermal junction
- Recall, too, that the epidermal cells of the basal layer are connected to the basement membrane via hemidesmosomes.
- The dermis contains lots of type 4 collagen.
- The superficial aspect of the dermis that is attached to the basement membrane of the epidermis is the lamina densa.
- The lamina densa of the dermis and the basement membrane of the epidermis are connected via anchoring filaments and anchoring fibrils.
- Note that anchoring filaments are composed of type 7 collagen.

[edit] Describe the cellular components of the epidermis and their functions
- There are four major cells of the epidermis: keratinocytes, melanocytes, langerhan cells, and merkel cells.
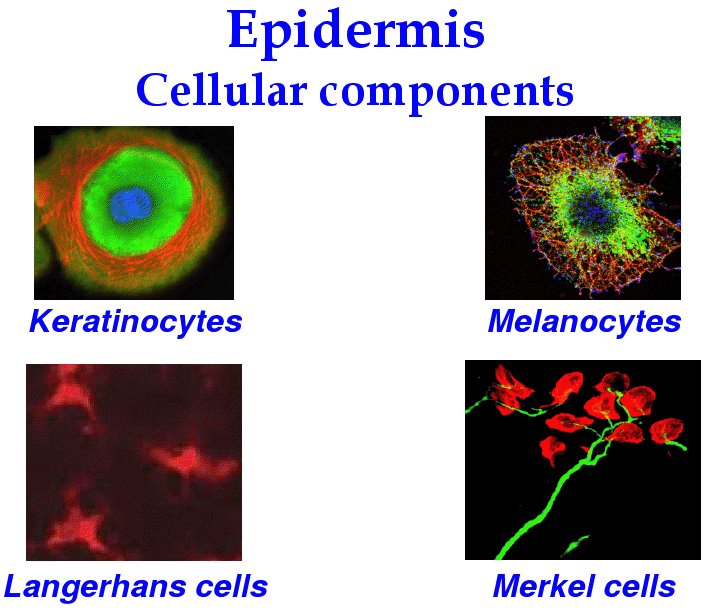
[edit] Keratinocytes

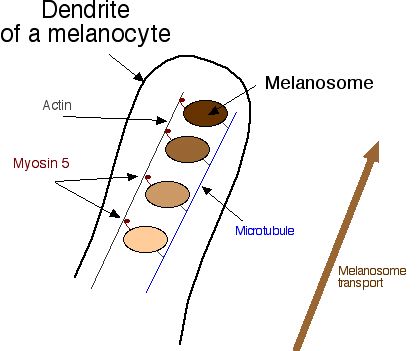
[edit] Melanocytes
- Melanocytes are derived from neural crest cells and function to generate the pigment melanin which protects cells from UV damage.
- Melanosomes are said to "mature" as they are produced; they turn from a light, circular shape to a dense cucumber shape.






[edit] Langerhans cells
- Langerhans cells reside primarily in the spinosum layer (think "spines and chinese are for killing bad guys!).


[edit] Merkel cells
- Merkel cells (like melanocytes) are found primarily in the basal layer, which makes sense because they are a sensation cell that needs to be near a nerve ending.
- Note that Merkel cells are found primarily in thick skin where touch needs to be highly sensitive.
- Merkel cells, along with the expanded terminal bulb of afferent, myelinated nerves, form the Merkel's corpuscle which detects touch as a mechanoreceptor.
- The Merkel cells contain dense-cored neurotransmitter granules.

[edit] Describe the structural organization of the dermis
[edit] Papillary layer of the dermis
- Connective tissue of the papillary layer is composed of type 1 collagen, type 2 collagen, and elastic fibers.
[edit] Reticular layer of the dermis
- The reticular layer is composed of type 1 collagen and regularly oriented elastic fibers (called Langer's lines).





[edit] Identify other structures in the skin
[edit] Vessels
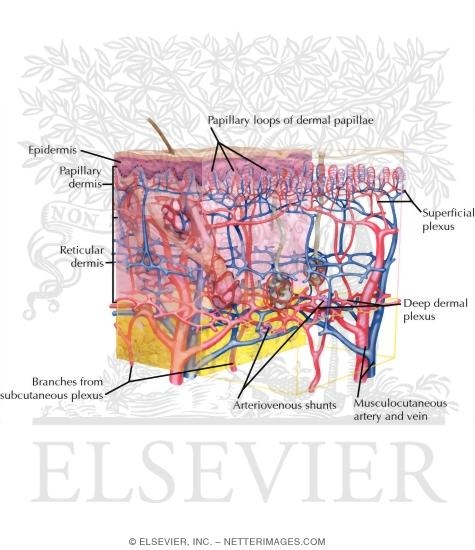

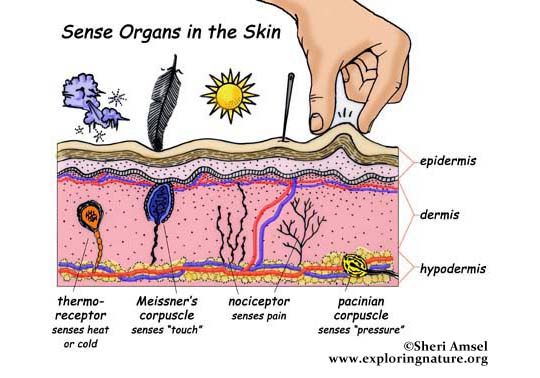
[edit] Sensory receptors
- There are four types of sensory receptors in the integumentary system (skin): free nerve endings, pacinian corpuscles, meissner's corpuscles, and ruffini's corpuscles.
[edit] Free nerve endings
- Free nerve endings are found in the stratum granulosum and detect fine touch, heat, and cold.


[edit] Pacinian corpuscles
- Pacinian corpuscles are nerve endings surrounded by an oval encapsulation of connective tissue in the deeper dermis and hypodermis.
- Think maraca shaped and all that vibration!






[edit] Meissner's corpuscles
- Meissner's corpuscles are found in the papillary layer of the dermis and are sensitive to low frequency stimuli.
- Doesn't "meissner" sound like an old miser with a low, grumpy voice?
- Meissner's corpuscles are shaped like tapered mitochondria and are oriented perpendicular to the skin.





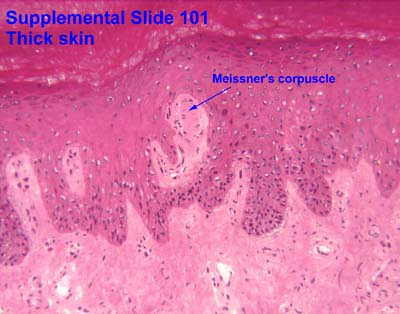


[edit] Ruffini's corpuscles
- Ruffini's corpuscles are simple mechanoreceptors and have an "elongated fusiform shape".

[edit] Sensory receptor images



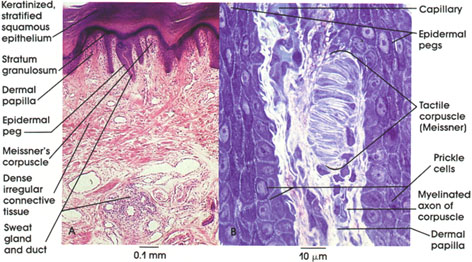


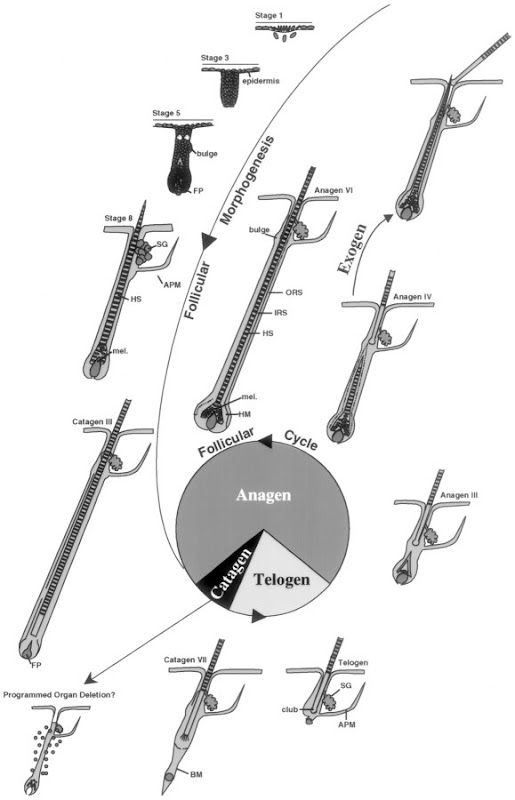
[edit] Hair follicles
- At the hair follicle a specialized layer called the glassy membrane (a type of basement membrane that is thickened and keratinized) separates the epidermis and the dermis.
- During hair growth, the follicle has a bulbous end at the deepest part.
- Hair has three layers (from outside in): cuticle, cortex, and medulla.
- The cuticle is the outer most and is comprised of squamous cells.
- The cortex contains cuboidal cells that differentiate into keratinized cells.
- The medulla contains large cells with vacuoles that are moderately keratinized.
- Sebum is released into the infundibulum which is a pilosebaceous canal that surrounds the base of the growing hair.





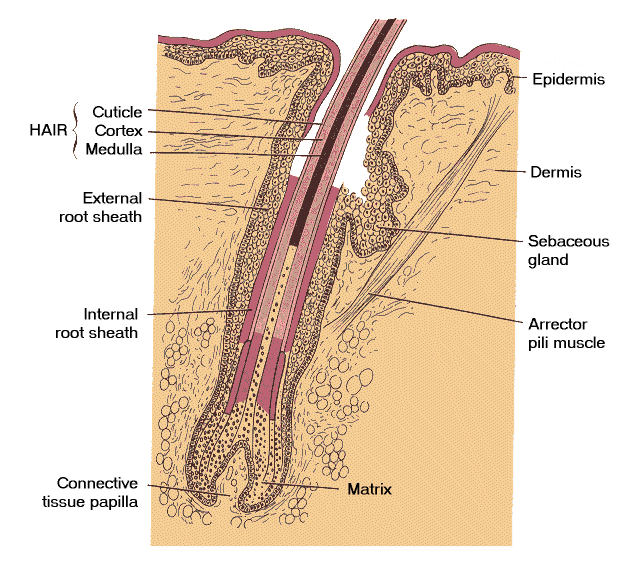


[edit] Phases of hair growth
[edit] Nails
- The nail plate sit in the nail bed which is formed by the stratum basale and spinosum.

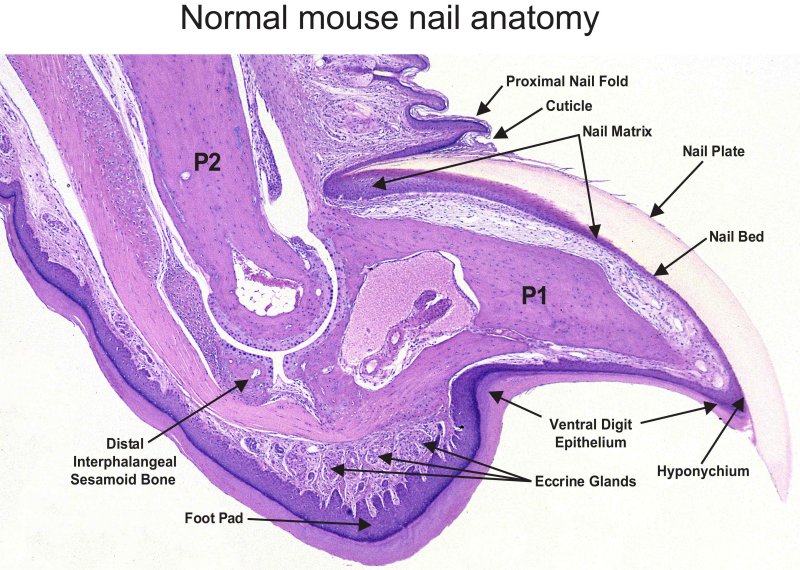
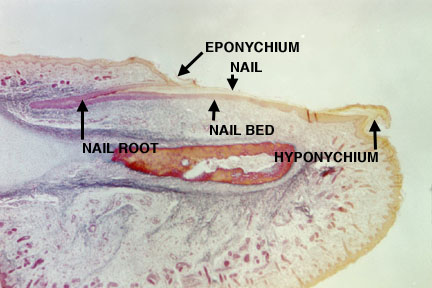


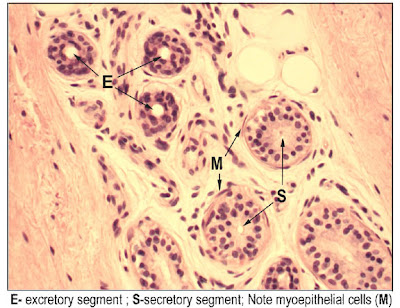
[edit] Glands

[edit] Sebaceous glands






[edit] Sweat glands
| Attribute | Merocrine | Apocrine |
|---|---|---|
| Secretion method | Merocrine | Apocrine and merocrine |
| Distribution | Widely distributed | Axillary and perineal regions only |
| Lumen size | Small lumen | Large lumen |
| Epithelial type | Stratified cuboidal | Simple cuboidal |
| Innervation | Cholinergic fibers (ach) | Adrenergic (cats) |
- Merocrine (eccrine):
- Apocrine:
- Visual differentiation:
- Lumen size: small = merocirne, large = apocrine
- Density: denser = merocrine, lighter = apocrine
- Surrounding cells: myoepithelial cells surround merocrine to help secrete, apocrine don't necessarily have surrounding cells.
[edit] Understand the mechanism of skin repair

[edit] Describe the histological findings in common skin diseases
[edit] Blistering
- Abnormalities at the epidermis-dermis junction are called bullous pemphigoid.
- Abnormalities of intercellular junctions are called pemphigus.
[edit] Psoraisis
- Psoriasis occurs when cells of the basal and spinosum layers demonstrate excessive proliferation and decreased cycle time which leads to increased thickness.
- One can identify psoriasis by the presence of nuclei in the stratum corneum; this finding is called parakeratosis.



[edit] Skin cancer

[edit] Basal cell carcinoma





[edit] Squamous cell carcinoma


[edit] Malignant melanoma

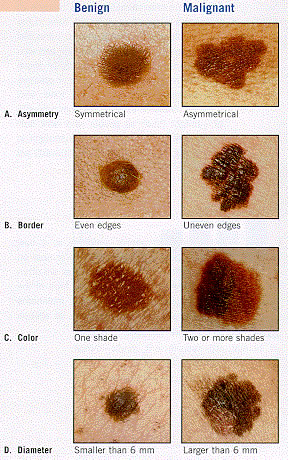


[edit] Griscelli syndrome
- Griscelli syndrome can result from a defective Rab27a protein which is part of the transport complex that moves melanosomes along microtubules for cyotocrine passage to other cells.
[edit] The eye
[edit] Optical anatomy

[edit] Wall of the eye

[edit] Eyeball function by component

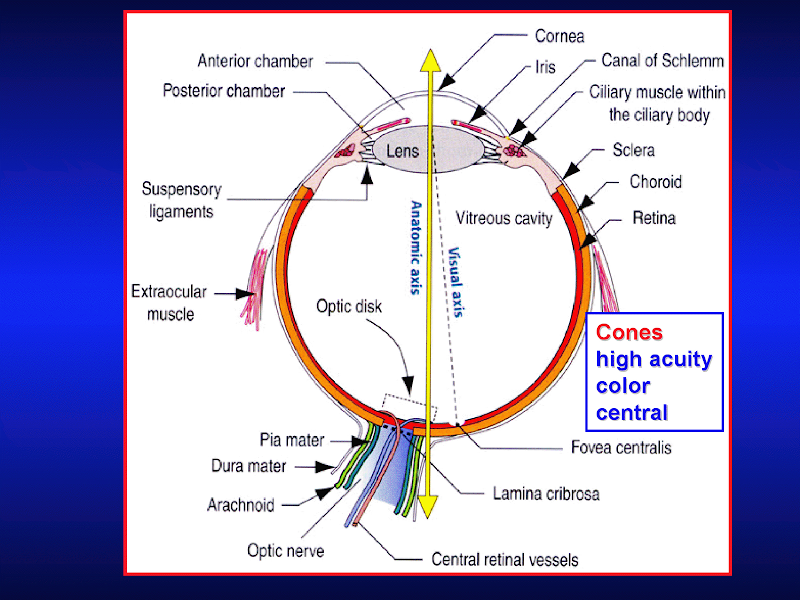
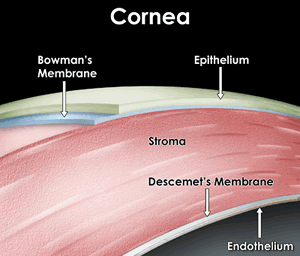
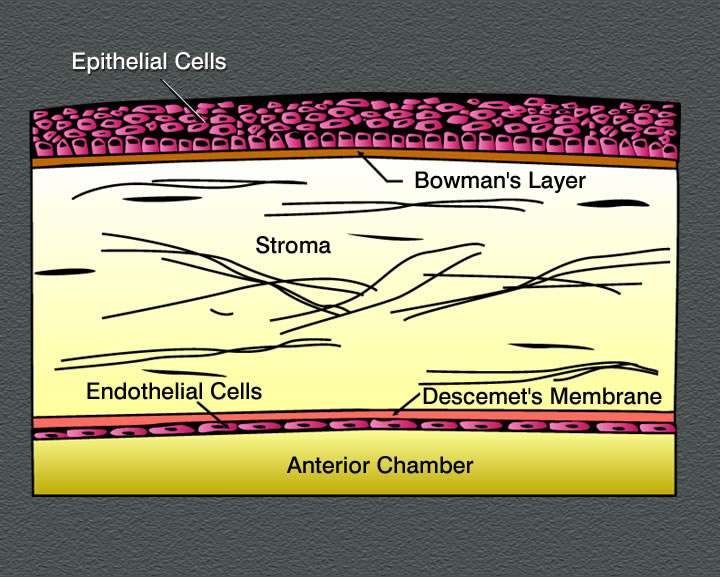
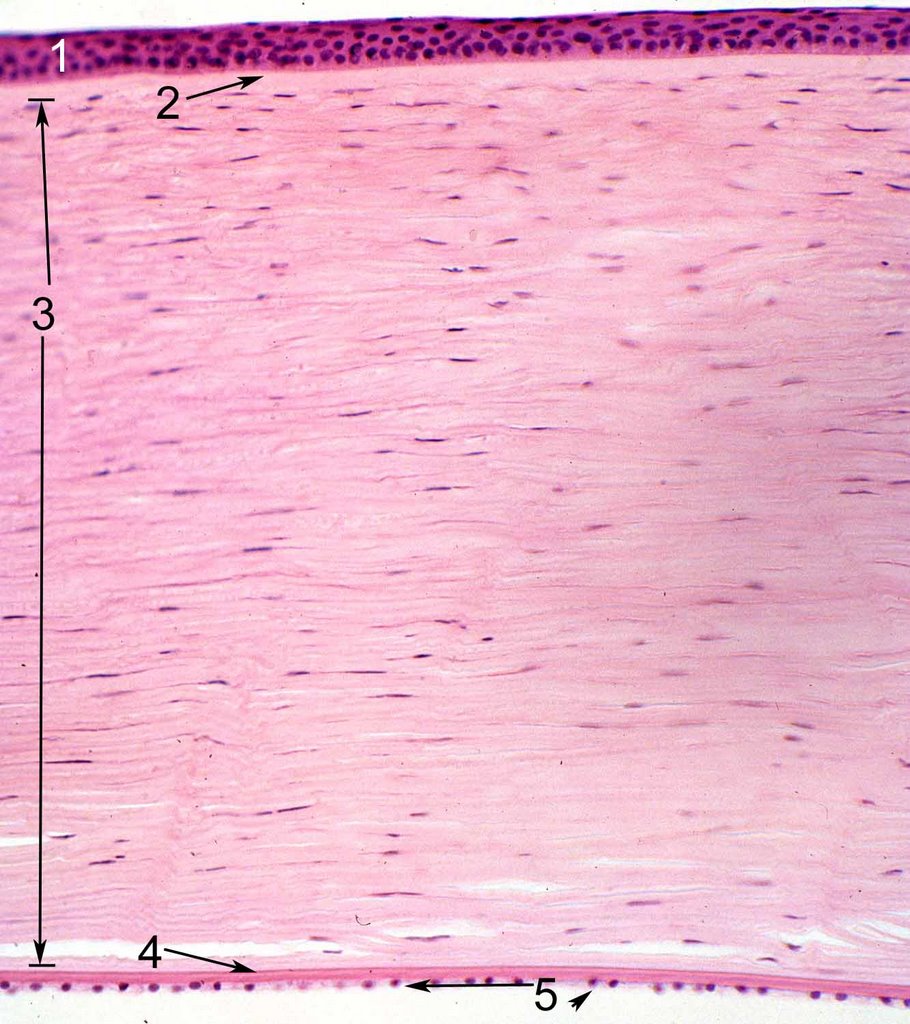
[edit] Cornea
- The layers (superficial to deep): epidermis, Bowman's membrane, stroma, Descemet's membrane, and endothelium.
- With age, Descemet's membrane decreases in transparency and leads to decreased light transmission.



[edit] Epithelium of the cornea
- The epithelium of the cornea is made of stratified, squamous, non-keratinized epithelium.



[edit] Stroma of the cornea
- The stroma is series of layers of fibrocytes, proteoglycans, and ECM fibers with alternately oriented collagen fibers.
- Note that collagen of the stroma is of type 1 and 5 and are non-fibrillar.

[edit] Endothelium of the cornea

[edit] Uvea

[edit] Choroid of the uvea
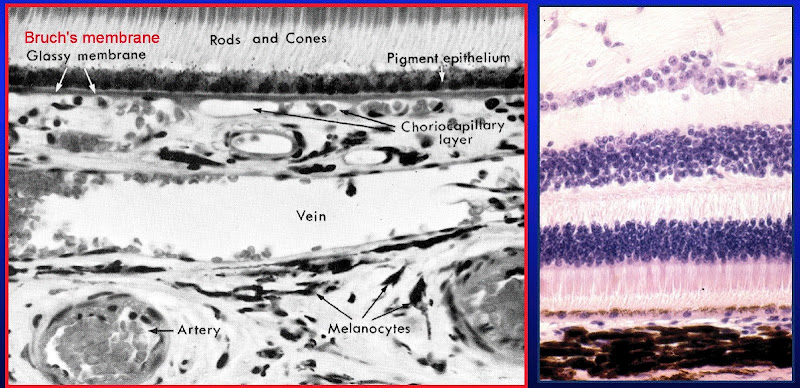
- The choroid is highly pigmented and found between the sclera (part of the tunica fibrous) and the retina.
- The choroid has three layers: sclera vasculature, retinal vasculature, and Brunch's membrane.
- The retinal vasculature layer is also called the choriocapillary layer.
- Bruch's membrane is also sometimes called a glassy membrane.

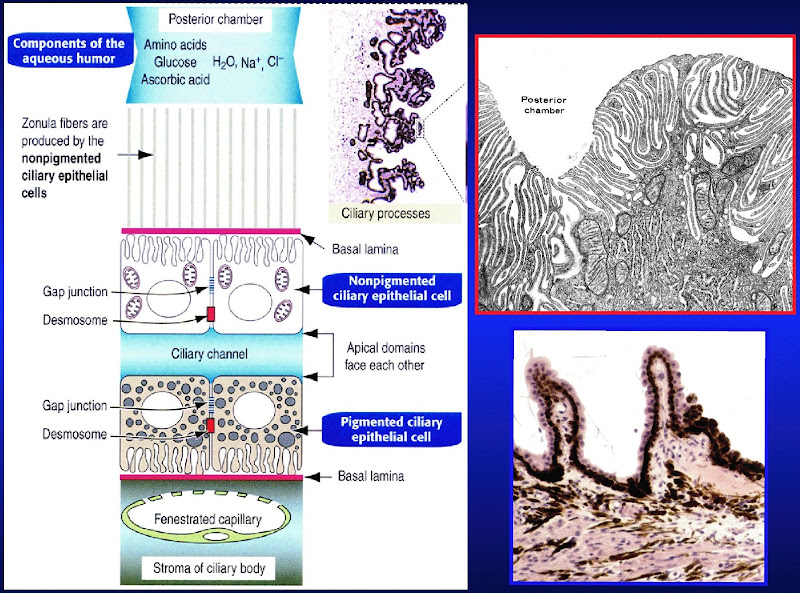
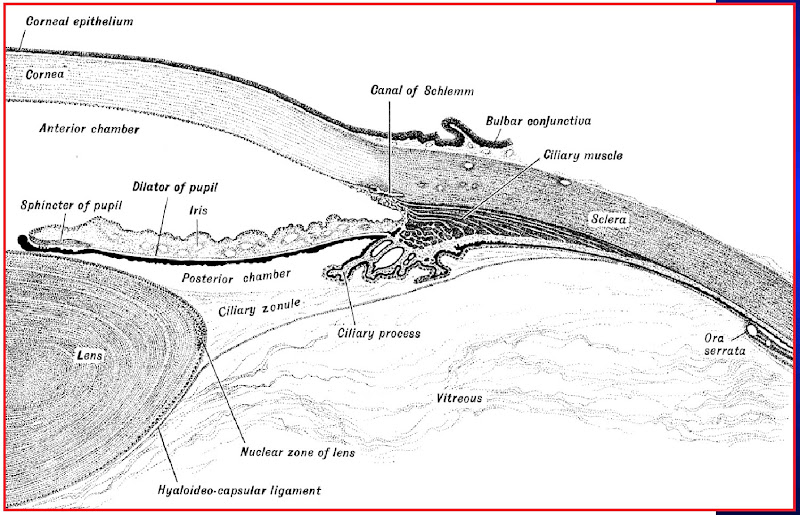
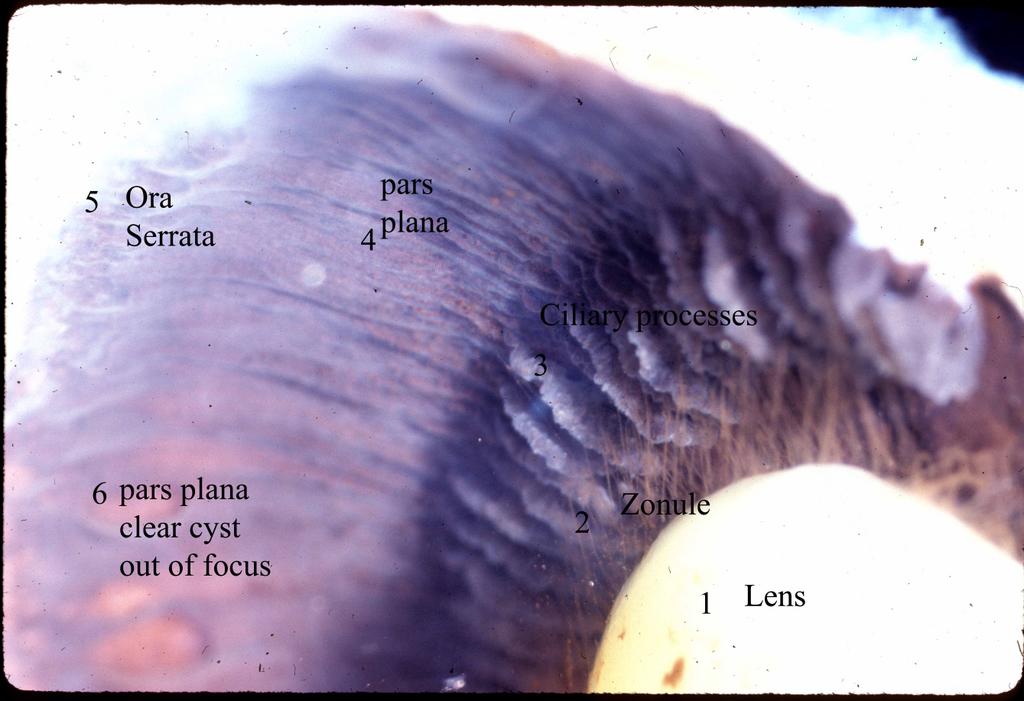
[edit] Ciliary body
- Far vision requires a flattened lens (think flat like a frisbee which you hope will go "far") so the ciliary muscle relaxes, the zonules tense, and the lens is pulled into a flatter shape.
- Near vision requires a bulged lens so the ciliary muscle contracts, the zonules relax, and the lens relaxes into a bulge.
- Oxytalin fibers are used to attach the basement membrane of the non-pigmented epithelium of the ciliary body (part of the uvea) to the basement membrane (called the lens capsule) of the lens.

- Ciliary processes:
- Aqueous solution has little protein, some glucose, and similar ion concentration as plasma.
- The pigmented layer (deep layer) of the ciliary process is continuous with the pigmented layer of the retina.
- The non-pigmented layer's (secretory, surface layer) apical surface faces the pigmented layer and the basolateral surface has lots of folds and borders the posterior chamber.
- We call the space between the pigmented and non-pigmented cells the ciliary channel and consider it a potential space.
- Blood-aqueous barrier: occluding junctions at the apex of the ciliary process's surface epithelium keeps blood and aqueous solution from mixing.

.jpg)
[edit] Iris
- Note that the posterior epithelium is one and the same as the myoepithelium: it has muscle fibers in it and is responsible for dilating the pupil.
- The malanocytes of the stroma determine eye color.
- The stroma is responsible for constricting the pupil.
- Note that dilation is sympathetic and contraction is parasympathetic.
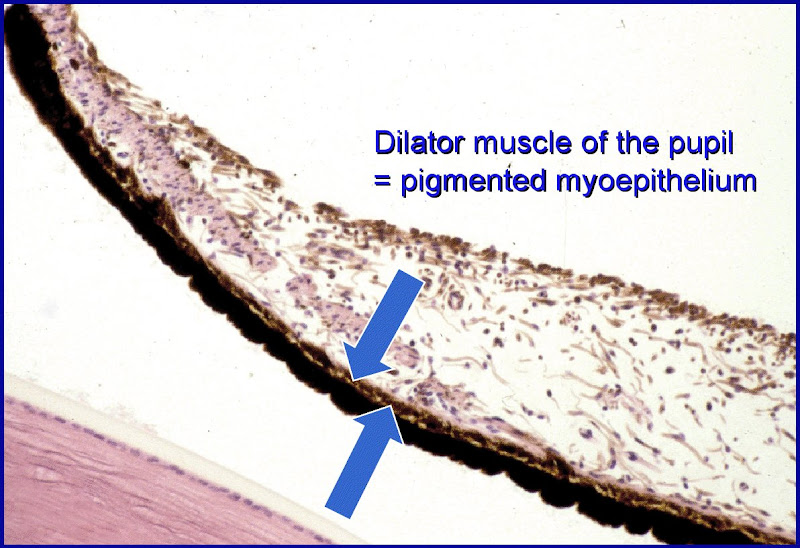
[edit] Eye color
[edit] Lens
- The epithelium on the anterior side of the lens is simple cuboidal.
- Note that the lens epithelium does not have occluding junctions.

- In the case of cataracts, lens fibers (the cells that span ant-post and have crystallin) turn opaque and refract light poorly.
- In the case of presbyopia, the lens loses elasticity (some loss is normal during aging) such that the pt cannot accomadate well and thus has poor near vision.
[edit] Retina
[edit] Pigmented epithelium of the Retina
- The pigmented epithelium of the retina is simple cuboidal and is adjacent to the inner-most layer of the uvea (Bruch's membrane).
- Note that secretion is enabled via Na-K ATPase.
- concentrates and estrifies vitamin A for easy reproduction of rhodopsin by rods
[edit] Neural retina


- Rods versus cones
- Rods use rhodopsin while cones have three distinct pigments for three colors: red, green, and blue.
| Attribute | Rods | Cones |
|---|---|---|
| Function | Night vision (scotopic), low acquity | Day vision (photopic), color, high acquity |
| Photopigment | Rhodopsin | 3 pigments, 3 wavelength specificities |
| Distribution | "Peripheral" | "Central" |
| Population count | 120 million | 6 million |